By Onoja Baba, Nigeria
The National Agency for Food and Drug Administration and Control (NAFDAC) has announced the recall of two substandard antibiotic products manufactured by Sam-Ace Ltd in Nigeria.
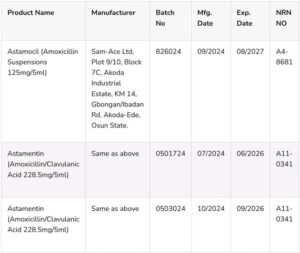
In a public alert (No. 34/2025), the agency identified the affected medicines as Astamocil (Amoxicillin Suspension 125mg/5ml), Batch No. 826024, and Astamentin (Amoxicillin/Clavulanic Acid Suspension 228.5mg), Batch Nos. 0503024 and 0501724.
According to NAFDAC, the products were discovered during a recent Risk-Based Post-Marketing Surveillance conducted at the Coordinated Wholesale Centre (CWC) in Kano. Laboratory analysis revealed that both suspensions failed quality tests due to low Active Pharmaceutical Ingredient (API) content, rendering them substandard.
The agency has directed Sam-Ace Ltd to immediately recall the affected batches from circulation to safeguard public health.
Amoxicillin is a penicillin-class antibiotic used to treat bacterial infections of the ear, nose, throat, genitourinary tract, skin, and respiratory system.
Its combination with Clavulanic Acid broadens its efficacy against bacteria that produce beta-lactamase enzymes, which can render amoxicillin ineffective.
NAFDAC urged healthcare professionals and consumers to remain vigilant and report any suspicion of substandard or falsified medicines and medical devices to the nearest NAFDAC office, via the toll-free number 0800-162-3322, or by email at sf.alert@nafdac.gov.ng (mailto:sf.alert@nafdac.gov.ng).
The agency reaffirmed its commitment to ensuring that only safe, effective, and quality medicines are available in the Nigerian market.
categories
recent posts


NIGERIA: Sowore Defends Himself In Court, Seeks Judge’s Recusal In Cybercrime Trial