NIGEIRA: NAFDAC Issues Public Alert On Recalled Multivitamin Supplements
By Ameenat Hamzat, Lagos, Nigeria
The National Agency for Food and Drug Administration and Control (NAFDAC) has issued Public Alert No. 023/2025 to notify Nigerians of a recall of certain multivitamin and iron supplements in the United States due to the risk of poisoning.
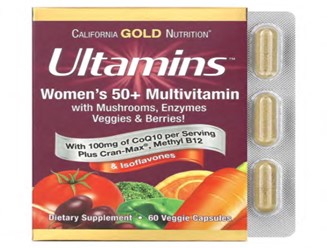
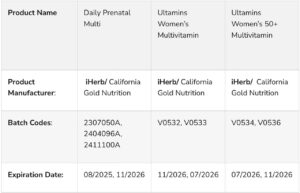
According to the agency in a statement posted on it’s official X handle, the affected products are marketed under the brand name CINCINNATI (WKRC) and include Daily Prenatal Multi, Ultamins Women’s Multivitamin, and Ultamins Women’s 50+ Multivitamin, all produced by iHerb/California Gold Nutrition.

According to the statement, it was stated that the recall report from the United States Consumer Product Safety Commission stated that approximately 60,000 units of these supplements were distributed through major retailers such as Walmart, Amazon, and Target.
The statement reads: ” The authorities discovered that the packages lack child-resistant packaging, which poses a risk of fatal poisoning if young children swallow the contents.”
NAFDAC however warned Nigerians to avoid those products, as they may be accessible to Nigerian consumers through online purchases or unregulated importation.
The agency also advised healthcare professionals and members of the public to report any suspected sale or distribution of substandard or falsified medicines to the nearest NAFDAC office, or through the following channels: Toll-free number: 0800-162-3322, email: sf.alert@nafdac.gov.ng
categories
recent posts

NIGERIA: FG Moves To Unlock 1,600MW Stranded Power With New Grid Company Plan

NIGERIA: Frank Mba, Seven DIGs Retire as PSC Promotes Replacements


NIGERIA: Google Expands AI Search Support To Yorùbá And Hausa

